In letters sent to manufacturers Olympus Corporation of the Americas, Pentax Medical and Fujifilm Medical Systems, U.S. Sen. Patty Murray (D-Wash.) addressed continuing concerns about infection transmission linked to duodenoscopes, according to an article on the Becker's Infection Control and Clinical Quality website.
"I have become increasingly concerned by the failure of Olympus to proactively warn patients and providers in the United States of the potential for infections," Murray said in the letter.
Government health officials have raised concerns over cleaning guidelines and procedures for the devices.
Sen. Murray demanded that manufacturers turn over copies of any cleaning guidance, safety alerts or warnings sent to hospitals or regulatory agencies in the past decade related to ERCP procedures.

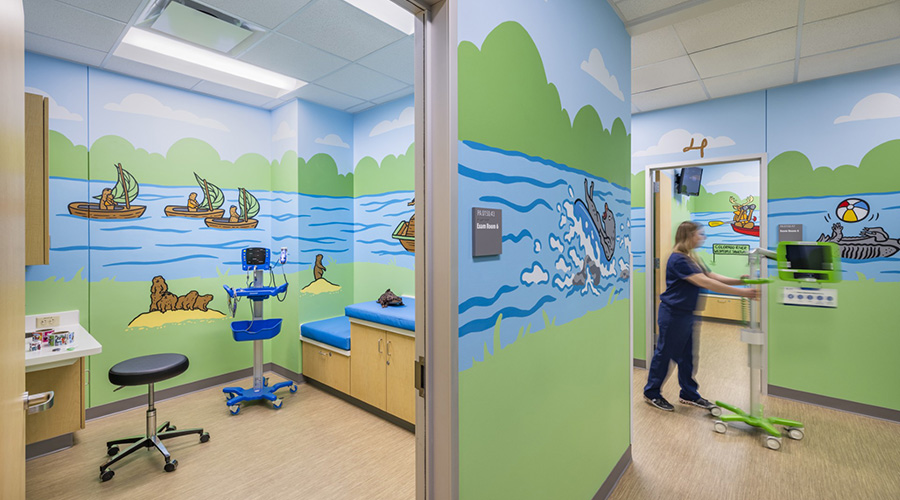 The Role of Positive Distraction in Pediatric Design
The Role of Positive Distraction in Pediatric Design Healthcare Waste is Fueling America's Debt
Healthcare Waste is Fueling America's Debt Prairie Lakes Healthcare System to Rebrand Following Sanford Health Merger
Prairie Lakes Healthcare System to Rebrand Following Sanford Health Merger How Digital Technologies Are Reshaping Performance in Healthcare Facilities
How Digital Technologies Are Reshaping Performance in Healthcare Facilities The Role of Plumbing in Healthcare-Associated Infections
The Role of Plumbing in Healthcare-Associated Infections